The Cancer Drug Testing Unit
Background:
- Established in August 2006.
- A systematic research unit for testing novel therapeutics in Asian prevalent cancer.
- 7 cancer cell line panels are ready for in vitro drug test: Hepatocarcinoma, pancreatic cancer, sarcoma, lung cancer, esophageal squamous carcinoma, cholangiocarcinoma, nasopharyngeal carcinoma.
- Drug resistance cell lines developed by CDTU: Lung cancer, esophageal squamous carcinoma, nasopharyngeal carcinoma.
- Cancer cell line derived xenografts in mice for in vivo drug efficacy test.
Mission and Goal:
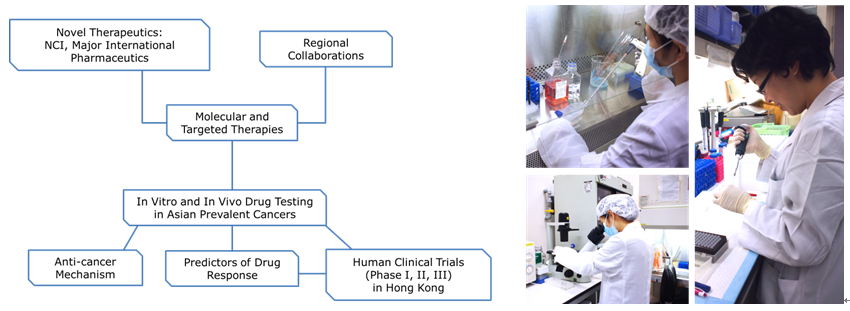
- To test in vitro and in vivo drug efficacy.
- To establish Asian cancer models for drug testing, attracting collaboration with international/regional pharmaceutics.
- To provide a preclinical platform for developmental/pipeline drugs to be tested in Asia-prevalent cancers in order to strengthen basic and translational oncology research in Asia.
Protocol Based Drug Test:

Stage I:
In vitro drug testing
Molecular Target Identification
Cytotoxicity
Cell cycle analysis
Apoptosis
Synergism with chemotherapeutic drugs or other anti-cancer drugs
Stage II:
In vivo drug testing (Only if efficacious in Stage I)
Dose optimization
Efficacy testing
Drug testing
Stage III:
In-depth Mechanistic Study Design
Tailor-designed studies for each drug according to the responses.
Recent publications:
| 1. Wong C.H., Ma B.B., Hui C.W., Tao Q., Chan A.T. Preclinical evaluation of afatinib (BIBW2992) in esophageal squamous cell carcinoma (ESCC). Am J Cancer Res 15; 5:3588-99. |
| 2. Wong C.H., Ma B.B., Cheong H.T., Hui C.W., Hui E.P., Chan A.T. Preclinical evaluation of PI3K inhibitor BYL719 as a single agent and its synergism in combination with cisplatin or MEK inhibitor in nasopharyngeal carcinoma (NPC). Am J Cancer Res 15; 5: 1496-506. |
| 3. Wong C.H., Loong H.H., Hui C.W., Lau C.P., Hui E.P., Ma B.B., Chan A.T. Preclinical evaluation of the PI3K-mTOR dual inhibitor PF-04691502 as a novel therapeutic drug in nasopharyngeal carcinoma. Invest New Drugs 13; 31: 1399-408. |


